Gallium (Ga), a toxic material, is produced as a by-product in both the zinc and aluminium production processes. Similarly, arsenic (As), which is also very toxic, is produced from ores such as AS2S3 or AS2S4. The process entails firstly oxidation of the ores to form AS2O3 and subsequently, through reduction with carbon, arsenic is produced.
In order to better appreciate the structure and the properties of gallium arsenide crystal, it is appropriate to focus some attention on the characteristics of the individual atoms themselves. The figure below shows Bohr’s model of the atomic structures for gallium and arsenic. Similar representation for silicon is also illustrated for comparison.
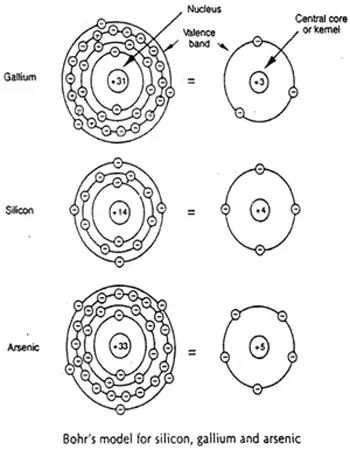
Bohr's Model For Si, Ga, and As
Electrons, travelling within their respective orbits, possess energy since they are a definite mass in motion (that is, rest mass of electron is 9.108*10-23gm). This means each electron in its relationship with its parent nucleus exhibits an energy value and functions at a distinct energy level. This energy level is dictated by the electron’s momentum and its physical proximity to the nucleus. The closer the electron is to the nucleus, the greater is the holding influence of the nucleus on the electron and the greater is the energy required for the electron to break loose and become free.
Outer orbit electrons are said to be stronger than inner orbit electrons because of their ability to break loose from the parent atom, and as a result they are referred to as ‘valence electrons’. The outer orbit in which valence electrons exist is called the ‘valence band’. It is the electrons from this band that are being considered in much of the discussions in the section to follow.
Crystal chemical bonds result through sharing of valence electrons. In materials such as Si, Ga and As, the outer-shell valence configuration can be represented by
Si→3s2 3p2
Ga→4s24p1
As→4s2 4p3
Here the core is not shown and the superscripts denote the number of electrons in the subshells (that is, s and p orbitals).
No comments:
Post a Comment